INTRODUCTION
The gold standard for achieving spinal fusion is still autologous iliac crest bone graft (ICBG), often associated with increased surgical time, postoperative pain, haematoma, infection, fracture of the ileum, neurovascular injury and cosmetic deformity [12]. In addition to minimally invasive (MI) approaches, inadequate local bone graft volume is problematic in revisions after a prior spinal decompression procedure. In order to avoid all of the above-mentioned, a host of bone graft alternatives have been developed [5,7,14,23-24]. Allografts possess minimal osteoinductive factors and lack osteogenic properties due to the processing to decrease antigenity [5,23-24]. They pose a potential risk for transmission of diseases, and incorporate slower and less completely with decreased vascularization and osteoconduction than autografts [23].
As synthetic, ceramic-based bone graft expanders, bioactive glasses (BAGs) are osteoconductive and osteostimulative, but non-osteoinductive materials that bond to bone without an intervening fibrous connective tissue layer [10,13,24]. They are composed mainly of silica, sodium oxide, calcium oxide and phosphates, and firstly form a hydroxycarbonate apatite layer on the surface of the bioactive glass, following initial dissolution [9,13]. Subsequently, BAG-bone bond formation occurs in stages by adsorption of biological structures on the glass surface, inflammation and macrophage action, and thereafter attachment and differentiation of stem cells [9]. As osteostimulative materials, BAGs stimulate recruitment and differentiation of osteoblasts, and activate them to produce new bone as a consequence of ionic dissolution [10,28]. Thereafter, generation and crystallization of bone matrix, and further bone growth occur more slowly than during the earlier stages [9]. Lastly, due to resistant surface of BAGs to cell-mediated degradation, physico-chemical degradation is only limited to the outer layer of a BAG granule, where dissolution and re-precipitation occur [21,36]. The rates of bioactivity and resorption of BAGs are dependent on the chemical composition, the surrounding pH, the temperature and the surface layers on the glass [24,35]. BAG S53P4 putty used in this study consists of a total of 60% by weight bioactive glass and is a synthetic, osteoconductive and osteostimulative paste-like bone void filler that was found biocompatible in our recent study [31].
Although previous studies have been carried out on BAG S53P4 granules in spinal fusion surgery with promising results, no clinical studies with the novel BAG S53P4 putty in spinal applications have been reported [6,20,30]. The primary aim of this study was to investigate the interbody fusion rate acquired with the mixture of BAG S53P4 putty and autologous local bone graft. Also, intervertebral cage subsidence is studied as well as radiological and clinical outcome.
MATERIALS AND METHODS
1. Study Design
A retrospective, single-surgeon and single-tertiary centre case study was undertaken to evaluate the novel, mouldable putty-form of BAG S53P4 as bone graft expander in an MI approach to transforaminal lumbar interbody fusion (TLIF) clinically. The enrolled patients had undergone unsuccessful conservative treatment of degenerative and/or postoperative back pain and/or radicular symptoms for at least a year before they were operated for lumbosacral MI-TLIF with BAG S53P4 putty as bone graft expander. This study was performed without contacting the patients. Permission from the hospital administration was obtained before the initiation of the study.
2. Patient Population
Twenty-one consecutive minimally invasive spinal (MIS) patients were included in the study. One of the twenty-one included patients died from an acute myocardial infarction nine days after the operation. The remaining twenty patients (13 female and 7 men) were operated on 24 lumbosacral levels with interbody fusions in the Department of Neurosurgery at Turku University Hospital between September 2014 and November 2016. One neurosurgeon (J.F.) operated on all patients. The mean age of the patients included was 49.3 years and five of them had previously undergone decompressive lumbosacral surgery. The patient demographics and perioperative diagnosis are presented in Table 1.
3. Bone Graft Expander
BAG S53P4 putty (BonAlive® putty; BonAlive Biomaterials Ltd., Turku, Finland) consists of 48 weight-% of osteostimulative calcium-phosphorous-sodium-silicate BAG S53P4 granules (size 0.5-0.8 mm) mixed with 12 weight-% of spherical (BAG S53P4) granules (size 0.09-0.425 mm) and 40 weight-% of a synthetic binder (mix of glycerol and three chain lengths of polyethylene glycols (PEGs)). The composition of the bioactive glass granules of BAG S53P4 putty is (by weight-%): SiO2 53%, Na2O 23%, CaO 20% and P2O5 4%. The binder serves as a temporary binding agent for the granules. The putty is provided as a premixed extrudable and mouldable, but cohesive material, packed in a syringe-like applicator and sterilized by gamma irradiation.
4. Surgical technique
All procedures were performed percutaneously on one side, and through a Wiltse’s approach on the symptomatic side. A navigation reference was first fixed with a clamp to the iliac crest. Then, cannulated Viper® MIS extended tab screws (DePuy Synthes, Le Locle, Switzerland) or in one case Everest® MIS screws (K2M, Leesbury, VA, USA) were inserted percutaneously using neuronavigation based on intraoperative 3D imaging (Stealth-Station® S7® Navigation System and O-arm® Imaging System, Medtronic Navigation, Louisville, CO, USA). Thereafter, a contralateral Wiltse’s approach was used. Subsequent to inserting the cannulated screws, facetectomy and discectomy were performed. Endplates were prepared and the fusion beds of the polyetheretherketone (PEEK) InterFuse® T-cage modules (VTI, Minneapolis, MN, USA) were filled with the mixture of BAG S53P4 putty and AB chips. After packing the mixture of BAG S53P4 putty and AB chips in the anterior disc space, and implanting of the cage, the rods were inserted, compression was applied and the set screws were tightened on both sides. 3D control imaging was performed, and a mixture of BAG S53P4 putty and AB chips was implanted on the transverse processes of the symptomatic side after scraping to bleeding bone, before closure in layers. The operation time, volume of bleeding, perioperative complications and postoperative hospitalization time were recorded (Table 1).
5. Radiological and Clinical Evaluation
Preoperative lumbar magnetic resonance imaging (MRI) and dynamic (flexion/extension) plain lumbar spine radiographs were carried out routinely. Intraoperatively, a 3D flat panel scan was performed in order to verify the correct position of the implants. Static plain radiographs were performed when ambulatory and at 3, 12 and 24 months. Patients with specific complaints were further investigated with CT and/or MRI scans.
The resorption of the mixture of BAG S53P4 putty and AB was estimated by plain lumbar spine radiography on a three-point scale (Table 2) by an independent neuroradiologist (J.H.). In grade 1, the columns containing the mixture of BAG S53P4 putty and AB were still distinguishable, while in grade 2 only partly distinguishable and in grade 3 not at all distinguishable. On the CT studies, interbody fusion was defined as presence of intervertebral bridging bone, without having had a revision or evidence of instrumentation loosening and/or breakage [11]. The same independent neuroradiologist assessed bridging bone on a four-point grading scale according to Table 3. The levels meeting the criteria of grades I or II were considered as bridged, whereas grades III and IV were judged not bridged. Moreover, fusion of the posterolateral implants was rated based on the Bridwell fusion grading system primarily from CTs, and if not available, from plain radiographs [2]. The disc height was evaluated from available preoperative and postoperative images.
The clinical follow-up in the outpatient clinic was at 3 and 12 months postoperative, the 24 months follow-up was merely a personal telephone interview. The minimum follow-up time was 12 months. The clinical outcome was measured as presence of low back pain, radicular leg pain, motor deficit, paresthesia and instability symptoms, and according to Odom’s criteria as excellent, good, fair or poor [27].
6. Statistics
Differences in resorption grades between two time points at a time were tested using the test of symmetry. The non-parametric Wilcoxon signed-rank test was used to measure the differences in disc heights before and after the operation. p-values less than 0.05 were considered statistically significant. Statistical analyses were carried out using SPSS system for Mac, version 24 (IBM Corp., Armonk, NY, USA).
RESULTS
Four patients were operated for two-level and 16 patients for single level MI-TLIF. A total of 24 MI-TLIF levels were operated in twenty patients. The most common operated level was L5-S1 (half of patients). A total of 58.4% of patients had either degenerative spondylolisthesis or spondylolysis and -olisthesis. The mean intraoperative bleeding was 292 mL (standard deviation (SD): 136 mL), the mean operation time 343 min (SD: 57 min) and the mean hospitalization time 4.0 days (SD: 1.7 days). Perioperative parameters are presented in Table 1.
The achieved interbody fusion could not be reliably estimated from static plain lumbar spine radiographs. Instead, the resorption of the mixture of BAG S53P4 putty and AB chips was graded according to static plain radiographs (Table 2). At three months postoperative, the above-mentioned mixture was still distinguishable in the fusion bed columns of the cage in 58.3% of the levels, whereas only in 8.3% at 12 months and in 5.3% at 24 months postoperative compared to immediate postoperative radiographs. In contrast, these columns were not distinguishable in 4.2% of the levels at 3 months, in 37.5% at 12 months and in 47.4% at 24 months postoperative, respectively (Table 4). The resorption was significantly more progressed in radiographs at 12 months compared with those at three months (p<0.001), but not in radiographs at 24 months compared with those at 12 months (p=0.135).
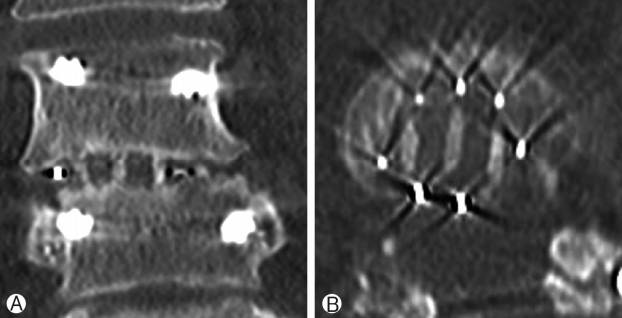
If indicated on clinical monitoring in the follow-up visits, patients were investigated with CT and/or MRI scans. Totally, ten CT scans were done for eight patients at 1-18 months postoperative (mean: 10.3 months, SD: 6.0 months). All CT scans were judged to show bridging bone across the intervertebral space. According to bridging bone criteria (Table 3), five of the nine investigated levels were graded as definitely fused (Fig. 1) and four levels as probably fused. The scans showing grade II (probably fused) were conducted at 1-18 months postoperative. One patient had cage dislocation of 2-3 mm posteriorly in static plain radiograph in L5/S1-level at three months postoperative and lucency around a sacral screw and breakage of the other sacral screw in CT at nine months postoperative. This level was graded as probably fused with bridging bone criteria. A total of 15 patients (75.0%) had posterolateral bone graft mixture unilaterally on the transverse processes and one bilaterally. Consequently, only one of the 17 levels showed Bridwell grade 1 fusion (fused with remodelling and trabeculae present), whereas the rest 16 levels were graded as Bridwell grade 4 (fusion absent with collapse/resorption of graft) in CTs and static plain radiographs at 12 months postoperative [2].
One patient had intervertebral cage subsidence/cage being inside the upper endplate and vertebral bone of 6 mm already in the intraoperative 3D flat panel scan. This subsidence remained unchanged until the latest follow-up radiograph at ten months postoperative. The mean disc height preoperatively was 6.3 mm (SD: 1.7 mm) and postoperatively 7.9 mm (SD: 1.2 mm) (Table 4). The median of difference of disc height was 1.0 mm greater postoperatively compared with that of preoperatively (p<0.001).
Postoperatively, two patients presented with new radicular pain, one with new motor deficit and two with new paresthesia. In contrast, 14 patients found relief of low back pain and radicular pain, eight of instability symptoms, five of motor deficit, four of paresthesia. According to Odom’s criteria, the patients’ clinical outcome was described at the latest clinical follow-up visit as excellent or good in 65.0% of the patients and poor in 20.0% of the patients. Clinical outcome results are presented in Table 5.
DISCUSSION
The interbody fusion rate of this study is 95.8% with the mixture of BAG S53P4 putty and AB assuming that patients not fused would be symptomatic within 12 months. Fifteen patients completed two-year follow-up without hardware failure. Two patients with new postoperative radicular pain, one patient with new L5 motor weakness and two patients with new paresthesia were further investigated with lumbar spine CT and/or MRI. One patient suffering from new radicular pain, had a clear hardware failure, despite his interbody fusion having been graded as probably fused. The patient with new motor weakness had a new disc herniation in the lower operated level, but the symptom relieved with conservative treatment. In another patient, a new paresthesia, due to an adjacent level disc herniation was relieved with conservative treatment. In two patients, no explanation for radicular pain and paresthesia was found. No postoperative infections were detected.
A systematic review suggests 83.4-100% fusion rates for MI-TLIF in degenerative spine, when using AB or allograft with or without rhBMP-2 [3]. In another systematic review, an average interbody bridging of 94.7% is presented for MI-TLIF with AB or allograft with or without recombinant human bone morphogenetic protein-2 (rh-BMP-2) [1]. By using a variety of different graft materials (AB, allograft cellular bone matrix, bone marrow aspirate, rh-BMP-2, corticocancellous chips, demineralized bone matrix (DBM)) alone or in combination in MI-TLIFs, interbody bridging was observed in 88% of the levels over 12 months and in 95% of the levels over 24 months [11]. In a recent meta-analysis, interbody bridging ranging from 91.8% to 99.1% was detected using combinations of AB with or without allograft and rh-BMP [29]. Interbody bridging of 96.6% and 92.5% were detected with and without rh-BMP, respectively [29]. The lowest interbody bridging rate was seen with isolated use of local AB (91.8%) and the highest by using local AB with bone expander and rh-BMP (99.1%) [29]. Further, the highest interbody bridging rate without the use of BMP was achieved with local AB and bone expander (93.1%) [29].
When comparing the achieved arthrodesis rates in MI-TLIFs between the cohorts of local AB and silicate hydroxyapatite ceramic bone graft expander (ActifuseTM) or local AB and rh-BMP-2, a radiographical arthrodesis of 65% in the Actifuse cohort and 92% in the rh-BMP-2 cohort were achieved [25]. Only in 26.7% of the levels solid fusion, in 34.1% indeterminate and in 38.6% inadequate fusion were assessed in PLIFs filled with β-tricalcium phosphate (β-TCP) and bone marrow aspirate (BMA) based on CT at 12 months postoperative [32]. In another study, using β-TCP with a resorbable polymer (ChronOSTM Strip) together with BMA and local AB in posterolateral fusion together with interbody support, interbody fusions of 54.1% and 71.2% were achieved in CTs at, respectively, 12 and 24 months postoperative [15]. Interbody fusion rates of 84.6% and 92.3% were estimated for a type 1 collagen/hydroxyapatite (HA) matrix (Healos®) with BMA, and AB, respectively, in PLIF from dynamic and static plain radiographs at 24 months postoperative [26]. Furthermore, (1) HA bone chip and local AB, (2) ICBG and local AB, and (3) local AB groups were shown to have 91.7%, 92.9%, and 94.6% fusion rates, respectively, in TLIFs evaluated by static plain radiographs at 12 months [16]. A mixture of local AB and bioactive apatitewollastonite granules containing glass ceramic yielded solid fusion in 92.0% of TLIF levels in static and dynamic plain radiographs at 6 months postoperative [8].
Subsidence of an interbody device in lumbar fusion is defined as sinking into one or both of the vertebral endplates, usually of ≥2-3 mm [4,18]. As a result of loss of disc height, subsidence can result in partial loss of ligamentous stability, in the return of foraminal stenosis and ensuing nerve root stenosis [19,33]. Particularly at the critical levels of L4/L5 and L5/S1, subsidence may also result in the loss of lordotic correction with consequent sagittal imbalance [22]. In our study, no subsidence was detected. According to a prior MI-TLIF study using a PEEK cage, the rates of cage subsidence of >2 mm and of >4 mm were 14.8% and 6.6%, respectively, at the last follow-up of 24-45 months postoperative [17]. The subsidence occurred in follow-ups on average at 7.2 months postoperative (SD: 8.5 months), and in all 1-25 months postoperative [17]. In another study on TLIF, subsidence of ≥10% in 52.9% of the allograft spacer with adjuvant rh-BMP-2 group and in 12% of the allograft/DBM group were seen [34].
Only one of 17 levels of posterolateral bone graft mixture of BAG S53P4 and AB led to formation of solid bony fusion. This may be due to shortage of stress to the remodelling posterolateral bone because of the well-supporting and load-bearing framework of interbody cage, and later interbody fusion [7].
The main limitations of the current study are its retrospective nature and the lack of a control group. Also, the sample size was small. Assessing interbody fusion from plain radiographs was unreliable, but due to potential harm from ionizing radiation associated with CT, these were restricted to symptomatic patients. Finally, pain, disability or other outcome scales, such as visual analogue pain scale or the Oswestry disability index, were not routinely in preoperative and postoperative use.
CONCLUSION
Novel BAG S53P4 putty as bone graft expander together with AB provides at least comparable results with previously tested bone graft expanders and fusion enhancers in achieving lumbosacral interbody fusion without subsidence. A prospective, randomized controlled study is needed to further analyse particularly the clinical outcome.