AbstractObjective There have been several reports of minimally invasive decompression for cervical canal stenosis and degenerative myelopathy. Most of these reports are for less than 4 levels and there have not been any comparative studies between Open and MIS cervical decompression for multilevel (≥4) degenerative cervical myelopathy.
Methods Twenty consecutive patients were allotted to undergo either ‘Open’ cervical laminectomy (n=10) or MIS posterior cervical decompression (n=10). All patients were evaluated for 1. Clinical, (JOA, MDI, NDI, Nurick grade, Blood loss, Duration of surgery); 2. Radiological (CSA of dural sac and Spinal cord, Muscle edema on post-op T2W MRI); 3. Laboratory (TLC, CRP, ESR, CPK) and 4. Physical (Isometric neck extensor muscle strength). Differences between Open and MIS groups were calculated with respect to above parameters.
Results The mean number of levels decompressed was 4.4 (range, 4–6). MIS group had significantly longer duration of surgery and lesser blood loss as compared to open group. The patients in open group were more disabled than MIS group pre-operatively, as evidenced by higher MDI and NDI. However, proportionate improvements were seen in both groups post-operatively in terms of all clinical parameters. Postoperative increase in CSA of spinal cord was also identical in both groups. Elevations in CRP and ESR were significantly higher in Open group post-operatively as compared to MIS group. Post-operative extensor neck muscle strength improved to a higher extent in MIS group as compared to open group though this was not statistically significant. No patient had any major post-operative complications.
Conclusion MIS posterior cervical decompression is safe and effective, can achieve similar extent of decompression and degree of clinical improvement as compared to open surgery. MIS has definite advantages of lesser blood loss, reduced tissue injury and better improvement in post-operative neck muscle strength as compared to open surgery.
INTRODUCTIONDegenerative cervical Myelopathy is one of the most common progressive conditions affecting the older age groups. It is characterized by a combination of pathological changes in the cervical spine, both anterior (Disc bulges and Osteophytes) and posterior (Facet arthritis and ligamentum flavum hypertrophy). Consequently, surgical procedures to address the condition have utilized both anterior and posterior approaches for relieving spinal cord compression and maintaining/ restoring spinal stability. A posterior approach is generally preferred in cases with >3 level involvement, lordotic/ neutral spinal alignment and patients not fit for a major anterior procedure.
Conventional (Open) posterior cervical approaches necessitate detachment of paraspinal muscles from their attachment to the ligamentum nuchae and spinous processes, and lateral retraction throughout the surgical period. In addition to the direct surgical trauma, this can result in ischemic or denervation injury to the paraspinal muscles, together resulting in post-operative paraspinal muscle atrophy, imbalance between the flexor and extensor group and chronic axial neck pain [1,2]. Several studies have demonstrated reduced incidence of paraspinal atrophy and axial neck pain after using muscle sparing approaches and preserving attachments to C7 and C2 spinous processes during a conventional approach [3-9].
Minimally invasive muscle splitting approaches, introduced for the lumbar spine, have the potential to significantly reduce muscle injury and preserve the posterior tension band [10]. Their superiority in minimizing blood loss, reducing the duration of hospital stay, enabling earlier return to work and improving functional outcomes as compared to conventional procedures has been shown in several studies [11,12]. Recently, several reports of extension of this technique to the cervical spine have appeared in the literature and have shown promising outcomes [13,14].
There have been several reports in the literature describing the technique of minimally invasive posterior cervical decompression techniques for cervical myelopathy [13,15-20]. Almost all of these have utilized the technique for limited levels (2–3 levels) and there have not been any comparative outcome studies between minimally invasive and conventional (open) technique. To the best of our knowledge, ours in the first study to include multilevel cervical canal stenosis for minimally invasive decompression (Minimum 4 levels) and prospectively compare the outcomes against an equal number of patients undergoing conventional (open) multilevel cervical laminectomy and decompression.
MATERIALS AND METHODSTwenty consecutive cases with at least 4 levels of secondary canal stenosis on MRI studies and progressive symptoms correlating with degenerative cervical myelopathy were selected for the study. Ten patients underwent Conventional (open) posterior cervical decompressive laminectomy (OPEN) and ten patients underwent minimally invasive (Tube assisted) cervical canal decompression by a technique described briefly below. All patients were operated under IONM (Intra-operative neuro-monitoring) with motor (MEP) and sensory (SSEP) evoked potentials.
1. Surgical Technique1) Conventional (Open) Decompressive LaminectomyWith the patient in prone and head stabilized on a skeletal pin fixation headframe in neutral position and under appropriate aseptic precautions, posterior midline incision was placed and deepened in avascular midline plane (between the lamellar layers of ligamentum nuchae up to the tip of the spinous processes of the lamina needed to be resected. The muscular attachments were subperiosteally dissected from either side of the spinous processes and lamina and retracted laterally with a self-retaining retractor. Full thickness lateral gutters were drilled on either side at the junction of lamina and facet and en-bloc laminectomy done. Hemostasis was achieved and wound closed in layers.
2) Minimally Invasive (Tube-assisted) Multi-level Cervical dDecompression TechniqueWith the patient in prone and head stabilized on a skeletal pin fixation headframe in neutral position and under appropriate aseptic precautions, a double incision technique was used to access multiple levels from C2-7. More commonly, the first incision was placed on side of approach 1 cm lateral to the midline co-axial to the C3-4 disc space and was used to decompress C3 and C4 levels (Figure 1B, C). If C2 needed to be decompressed, the incision was placed co-axial to the C3 vertebral body and used to decompress C2 to C4 levels. A separate second incision was placed on the same side co-axial to the C5-6 disc space and used to decompress C5 and C6 levels (Figure 1F, G). If C7 needed to be decompressed, the tube could be angulated inferiorly with the same incision. The entire surgery was done with a 18 mm tubular retractor under the microscope. At each level, ipsilateral decompression was done and tube angulated to contralateral side to achieve contralateral decompression (similar to technique used in lumbar area) (Figure 1D). The detailed surgical technique is not described here.
2. Data Collection1) Clinical DataApart from the regular demographic information, VAS score for neck pain and arm pain was collected for each patient. The extent of disability and degree of myelopathy were noted by means of Nurick grade, Myelopathy Disability index (MDI) and Japanese Orthopedic Association (JOA) scores. In addition, Neck disability index (NDI) score was also collected. All the above clinical parameters were collected pre-operatively, POD 7, at 6 weeks and at 3 months.
2) Radiological DataAll patients underwent pre-operative dynamic radiographs to r/o any instability. Magnetic Resonance imaging (MRI) with all relevant sequences and CT scan of the cervical spine (occiput to D2) was done in all patients pre-operatively and on POD 2. On both MRI and CT, routine observations regarding alignment of the spine, levels of involvement, compression of the spinal cord, any intrinsic changes within the cord were observed (Figure 2). The important parameters which were included for analysis are the following: (1) Cross-sectional area – Dural sac – measured pre-operatively and post-operatively (Figure 3); (2) Cross-sectional area – Spinal cord – measured pre-operatively and post-operatively (Figure 3); (3) Post-operative signal change on T2W axial MRI image (Figure 4) – measured separately for Superficial group (paraspinal muscle, PSM) and Deep group (Semispinalis cervicis, SSC) and graded as 1, 0%–25%; 2, 26%–50%; 3, 51%–75% and 4, >75% of cross-sectional area of the muscle group affected. Measurements were taken separately at each disc level from C2-3 to C6-7.
3) Laboratory (Biochemical) DataBiochemical response to tissue injury was assessed by observing C-reactive protein (CRP), Erythrocyte sedimentation rate (ESR), Total Leucocyte count (TLC) and Creatine Phosphokinase (CPK) levels and trends following surgery in all patients. All the above parameters were assessed pre-operatively, POD 1, POD 3, POD 5 and on POD 7.
4) Physical DataEvaluation of maximum Isometric contraction strength of Neck Extensor Muscles was done using a pressure biofeedback device (Figure 5). The initial cuff pressure of this device was set at 40 mmHg. The patient was in supine lying with the cuff of the device placed below the external occipital protuberance. The patient was asked to lie down on a hard plinth in relaxed supine position ensuring normal cervical lordosis after the device was placed. The patient was advised to push the cuff down with isometric contraction of the neck extensor muscles without contracting the shoulder girdle muscles. Then the change in grade was recorded. Three movements with 2 minutes rest between them were repeated and the averages of obtained scores were recorded as the maximum isometric contraction strength of neck extensor muscles. These measurement were recorded for all patients in both groups pre-operatively and at 6 weeks following surgery.
3. Statistical AnalysisStatistical analysis was performed using SPSS software (2015, version 23.0; IBM Corp., Armonk, NY). Descriptive statistics with median values and interquartile variations were calculated and tabulated as shown below. Intergroup variations between MIS and Open groups were analyzed using non-parametric longitudinal design tests (Mann-Whitney U and mixed ANOVA).
RESULTSOf the twenty patients, 10 underwent conventional (open) laminectomy ad decompression (henceforth referred to as “Open” group) while the other ten underwent Minimally invasive decompression (henceforth referred to as “MIS” group). The most common levels decompressed were C3-6 in both MIS and open groups. The most common number of levels decompressed was 4 levels in both groups, which was also the minimum number of levels decompressed in any patient (Figure 6). There was no post-operative neurological deterioration in any patient. One patient in the open group had delayed wound healing with no long-term consequence. There were no complications in any other patient.
As enumerated in the Methods section, Results will be discussed under the following sections: 1. Clinical; 2. Radiological; 3. Biochemical; 4. Physical.
1. Clinical Data – Results (Table 1)The median duration of surgery was significantly longer for MIS group at 190 minutes compared to the open group which was 107 minutes. On the other hand, blood loss was significantly less in the MIS group with a median value of 150 mL as compared to open group in which it was 250 mL (Figure 7).
The extent of disability and myelopathy was more pronounced in the open group pre-operatively, as evidenced by the higher pre-operative median MDI, NDI and Nurick grades and lower JOA scores as compared to the MIS group, with the difference reaching significance for MDI and NDI (Table 1). Post-operatively, proportionate improvements were noted in both MIS and open groups in all the above parameters (Figure 8). The pre-operative difference of higher MDI, NDI, Nurick and lower JOA scores in open group was maintained in the post-operative period at 7 days, 6 weeks and 3 months follow-up periods, though the margin of difference reduced for all parameters post-operatively. While MDI maintained significant difference between open and MIS groups at 7 days and 6 weeks post-operative and lost significance at 3 months post-operative follow-up, significance was lacking for the other parameters (JOA, NDI, Nurick grade) at all post-operative points of evaluation. To enumerate, though patients in open group were more disabled pre-operatively than those in MIS group, proportionate improvements were seen in both groups post-operatively with no significant difference between the groups at 3 months with respect to any clinical parameter assessed.
2. Radiological Data – Results (Table 2)There was no noticeable difference in the CSA of the dural sac or spinal cord between the MIS and Open groups pre-operatively. As expected, there was significant improvements in CSA of both dural and spinal cord post-operatively in both groups (Figure 9). The dural sac CSA increased more in the open group as compared to MIS group, which could be explained by the inherent nature of the surgery where entire laminectomy was performed in open group while only a hemilaminectomy and decompression was performed in MIS group. However, this difference was not statistically significant. More importantly, what mattered was that improvements in CSA of spinal cord remained proportional with no noticeable difference between the two groups (Figure 9B). The change in the CSA of dural and spinal cord was also calculated for each level independently. While dural sac dimensions increased more in the open group as compared to MIS group at all individual levels (Figure 9A), there was no significant difference at any level with respect to change in CSA of spinal cord or dural sac between pre-operative and post-operative values.
Post-operative signal changes in the muscles assessed on T2W axial MR sequences were evidently more prominent in the open group as compared to MIS group. Statistically significant differences were obviously found on the side contralateral to entry in MIS group as compared to open group, since there was no muscle dissection at all on that side in MIS group as compared to open group wherein bilateral muscle groups were dissected from their attachments and retracted for performing laminectomy. More noticeably, even on side ipsilateral to entry, MIS resulted in significantly lesser muscle signal changes at peripheral levels (C2-3 and C6-7) in the deep group (semispinalis cervicis, SSC) as compared to the open group (Table 3).
3. Laboratory Data – Results (Table 4)There were no significant pre-operative differences between the open and MIS groups with respect to TLC, CRP, ESR or CPK. Post-operatively, the elevations in CRP levels showed marked difference between open and MIS groups, with open group having significantly higher elevations in CRP as compared to MIS group, remaining statistically significant till 7th post-operative day (Figure 10). Elevations in TLC and CPK were higher in open group as compared to MIS group, but were not statistically significant. Unexpectedly, ESR elevations were also significantly higher in open group as compared to MIS group from first until the fifth post-operative day.
4. Physical Data – Results (Table 5) (Figure 11)Changes in isometric extensor muscle strength were calculated with a biofeedback mechanism as described in methods section. Compared to pre-operative levels, muscle strength significantly improved post-operatively in both open and MIS groups. The degree of improvement was higher in the MIS group (27.5%) as compared to open group (15.7%). However, this difference did not reach statistical significance.
DISCUSSIONThere have been several reports of minimally invasive posterior cervical decompression for degenerative myelopathy in the literature in the last 2 decades with an inconsistent frequency. In an early reported series of 13 patients, Boehm et al. [16] reported on 9 patients who underwent interlaminar decompression through a tube for myelopathy with most of the patient in their series being only single level, basically using this procedure as an alternative for anterior cervical discectomy. Using a tube assisted technique similar to the one used in our series, Santiago and Fessler reported adequate decompression at 6 of the 8 levels decompressed in 4 patients [13]. Hur et al. [17] also used a tube assisted technique to decompress 13 levels in 6 patients, with a maximum of 3 levels in any patient and reported good outcomes in all their patients. Hernandez et al. [18] described a 10-step technique to report the safety and efficacy of minimally invasive cervical decompression with a tubular retractor in 15 patients, with majority being single level decompression (range, 1–3). There have also been several reports of endoscope assisted decompression for cervical canal stenosis, all of them restricting to 3 levels of decompression with the majority patients undergoing one or 2-level decompression [15,20,21]. To the best of our knowledge, there are no reports of minimally invasive techniques being employed in decompressing 4 or more levels in the cervical spine.
Comparative studies between ‘MIS’ and ‘Open’ posterior cervical decompression are the only effective means to ascertain the safety and benefits of a technically challenging MIS cervical decompression as compared to the much simpler, more widely practiced technique of ‘Open’ posterior cervical laminectomy. To the best of our knowledge, ours is the first and only study so far to prospectively and comprehensively compare and report the differences between ‘Open’ and ‘MIS’ posterior cervical decompression. Abbas et al compared the outcomes between anterior fusion surgery (ACDF, 29 patients) with posterior minimally invasive cervical decompression (45 patients, Mean 2.8 levels) and reported similar outcomes in both groups [19].
In the course of the study, the authors aimed to answer these three primary questions: 1. Does MIS achieve adequate decompression as Open?; 2. Is the extent of clinical improvement same in MIS and Open techniques?; 3. Are there benefits of MIS technique compared to Open?
1. Does MIS Achieve Adequate Decompression as Open?This was the primary and the most important question the authors aimed to answer in the present study. Though there are several reports of MIS posterior cervical decompression in the literature, none of them had compared the extent of decompression achieved with respect to radiological parameters and had reported on improvement in clinical variables alone [13,15,16,18]. In the present study, improvements in CSA of spinal cord (the factor directly representing adequacy of decompression) were similar in both open and MIS groups. The dural sac CSA improved more in ‘open’ group as compared to ‘MIS’ group, understandably due to a wider laminectomy than the unilateral laminotomy of MIS procedure. Since the spinal cord CSA improved proportionately in both groups, the additional increase in dural sac CSA offered by ‘open’ laminectomy was probably not clinically relevant. It is hence safe to infer that adequacy of spinal cord decompression in MIS is similar to that of open technique.
2. Is the Extent of Clinical Improvement Same in MIS and Open Techniques?The patients in the open group were more disabled pre-operatively as compared to those in MIS group, as evidenced by the intergroup pre-operative differences in MDI, JOA and Nurick grades. This is an inherent bias in case selection, presumably because the authors were reluctant in the earlier part of the study to select cases which were more disabled for the newer, yet unproven MIS technique. However, significant clinical improvement was seen in both groups post-operatively, with patient in open group continuing to exhibit more disability than patients in MIS group, though statistically insignificant. Notwithstanding the pre-operative selection bias, patients in both groups showed significant clinical improvement post-operatively.
3. Are There Benefits of MIS Technique Compared to Open?The authors compared several parameters – biochemical, radiological and Physical – to ascertain the benefit, if any, of MIS technique over its well-established counterpart, the open technique. Biochemical markers of tissue injury and acute phase reactants (CRP) showed significantly higher post-operative elevations in open group which was sustained for longer periods as compared to MIS technique. Post-operative muscle edema was significantly lesser in MIS group, obviously on the side contralateral to entry in MIS and at peripheral levels on the ipsilateral side as well. Post-operative isometric neck extensor strength improved by a better margin in patients who underwent MIS as compared to those underwent open decompression, though the difference was not statistically significant. Even with a small sample size, the above findings suggest considerable benefits for MIS procedure as compared to an open technique.
4. Limitations of the StudyThe authors understand the limitations of the present study. It’s a small group comparison and may not be an adequate sample to conclusively prove or disprove the observations of the study. A larger group with prospective analysis of all above sample is the need of the hour. Also, the follow-up period is limited to 3 months, primarily since the main objective of the study was to prove the adequacy of surgical decompression, extent of clinical improvement, and safety of MIS posterior cervical multilevel decompression as compared to open cervical decompression. Long term follow-up with documentation for sustained clinical improvement and benefits of MIS technique will conclusively answer the same questions over a long term.
CONCLUSIONDespite certain limitations, the present study is the first to prospectively and comprehensively compare ‘MIS’ and ‘open’ posterior multilevel (≥4) cervical decompression. Minimally invasive posterior multilevel cervical decompression for degenerative cervical myelopathy is a safe and effective technique, that can achieve similar extent of spinal cord decompression and degree of clinical improvement as a conventional ‘open’ posterior cervical decompression, in the form a laminectomy, can achieve. MIS cervical decompression has obvious benefits of reduced blood loss, less tissue injury and better post-operative extensor muscle function as compared to open laminectomy.
Figure 1.Illustrative case example demonstrating the 2-incision technique (H) used for multilevel posterior cervical MIS decompression. In this case, the upper incision has been used to decompress C3 (B) and C4 (C), while the lower incision has been used to decompress C5 (F) and C6 (G). (A, E) It represents the initial docking site and direction of the first dilator. (D) It shows appearance of decompressed dural sac at one level. 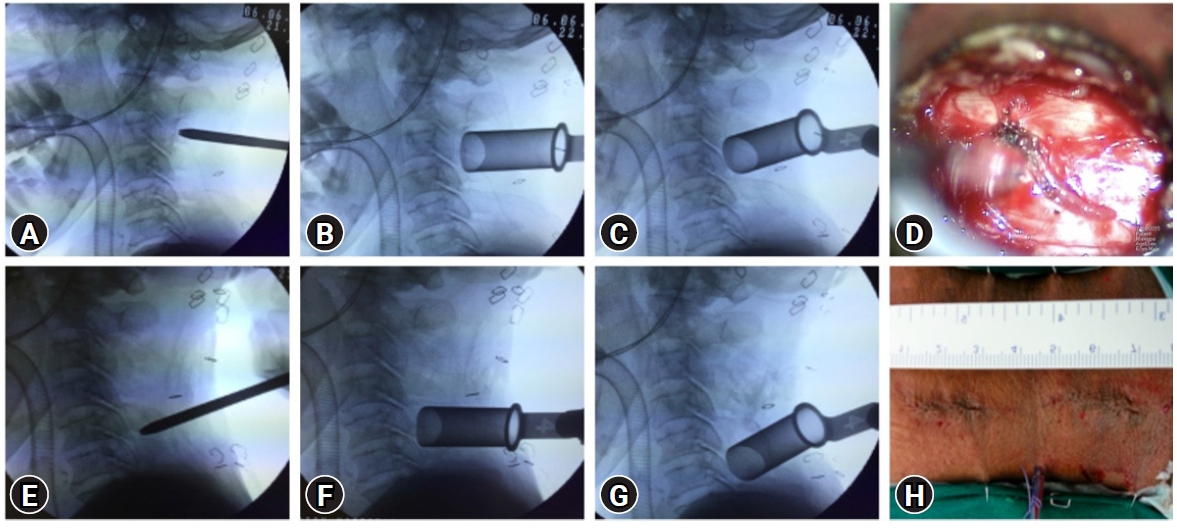
Figure 2.Illustrative case example showing secondary canal stenosis from C3-5 due to continuous OPLL (Ossified posterior longitudinal ligament). (A) Pre-op and (E) post-op mid sagittal T2W MRI image. (B) Pre-op and (F) post-op mid sagittal CT scan image. (C) Pre-op and (G) post-op Axial T2W MRI image. (D) Pre-op and (H) post-op Axial CT scan image. 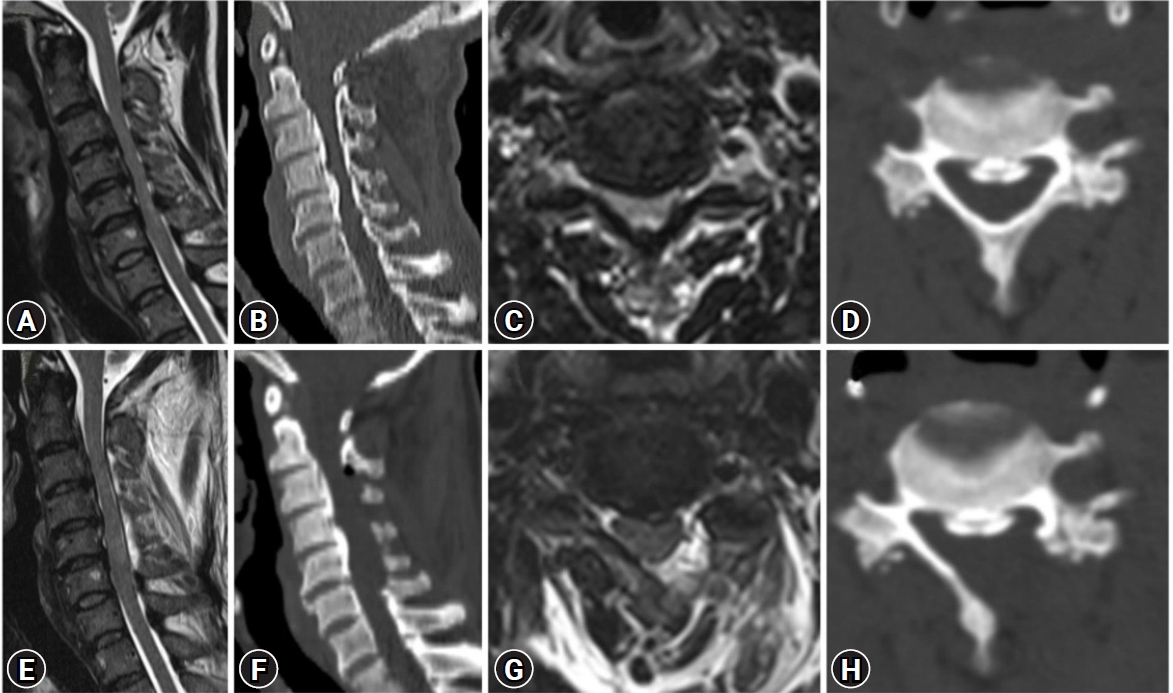
Figure 3.Illustrative case example showing the technique used for measuring CSA of Dural sac and spinal cord pre-operatively (A), post-operatively after open decompressive laminectomy (B), and after MIS decompression (C). 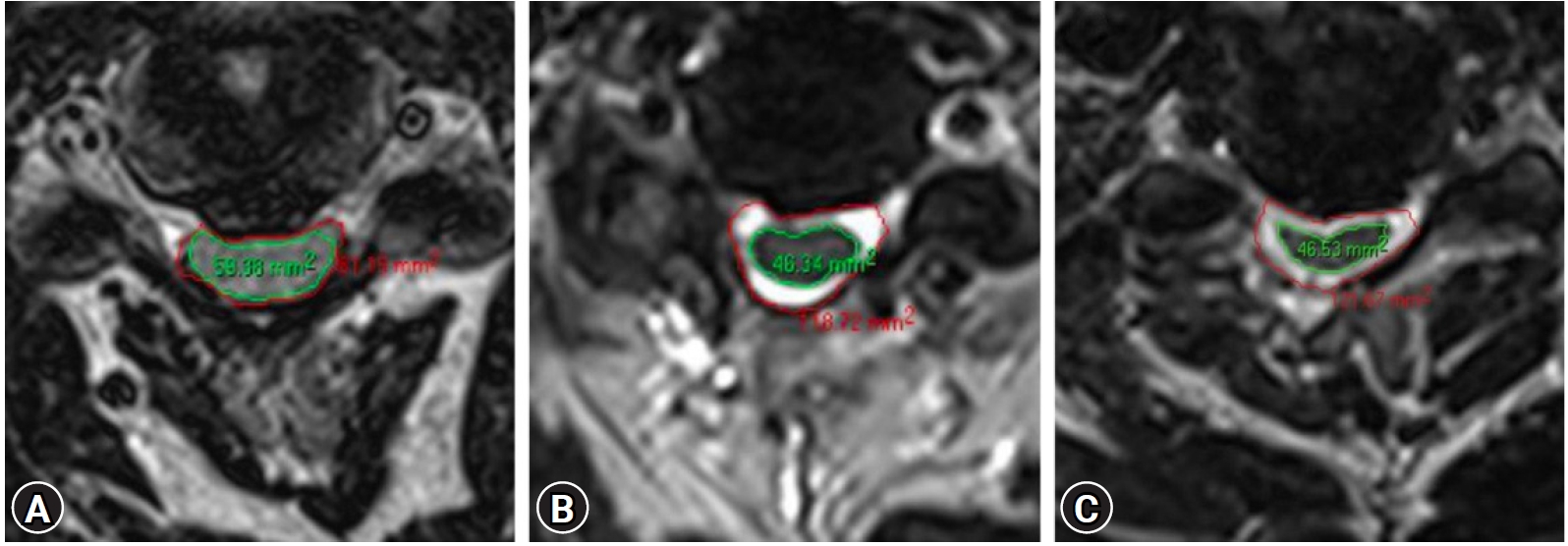
Figure 4.Illustrative case examples showing technique of measuring post-operative muscle edema on T2W axial image after open decompressive laminectomy (A) and MIS decompression (B). (C) Post-op image after MIS decompression showing minimal edema (<25%) on side of entry (left). (D) Post-op image after MIS decompression in a different case showing larger area (>75%) on left side (side of entry) and no changes on contralateral (right) side. 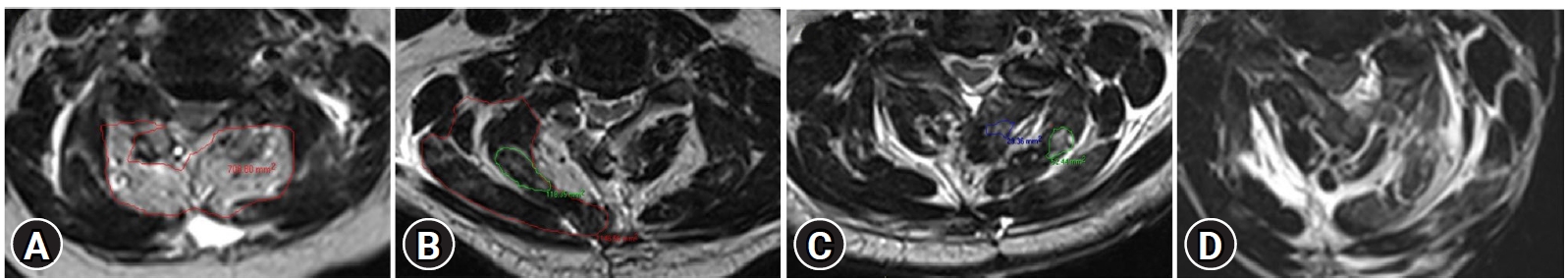
Figure 5.A volunteer demonstrating biofeedback device used to measure posterior neck muscle strength. 
Figure 6.Histograms depicting the details of levels decompressed (A) and median number of levels decompressed (B) in Open and MIS groups. 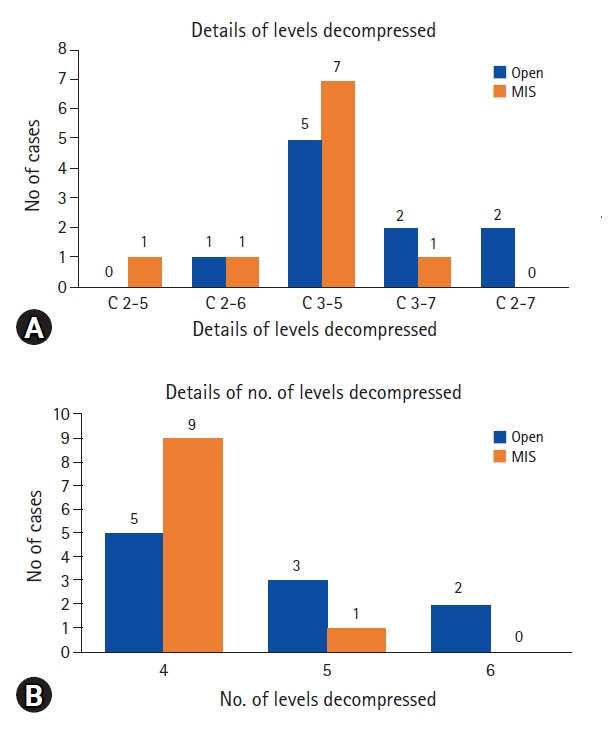
Figure 7.Histogram depicting comparison of duration of surgery and Intra-operative blood loss between Open and MIS groups. 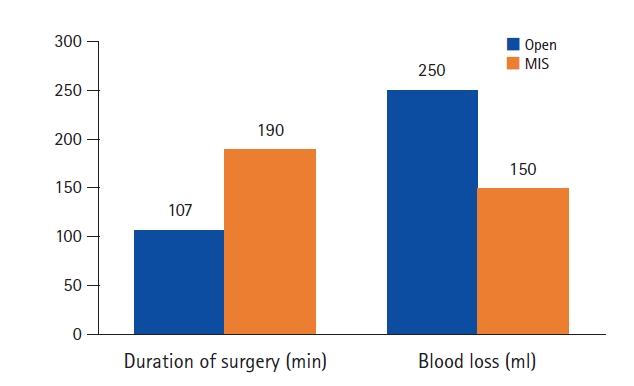
Figure 8.Line charts illustrating the comparison of pre-operative values and post-operative trends at various time points between Open and MIS groups with respect to JOA (A), MDI (B), NDI (C), and Nurick grade (D). 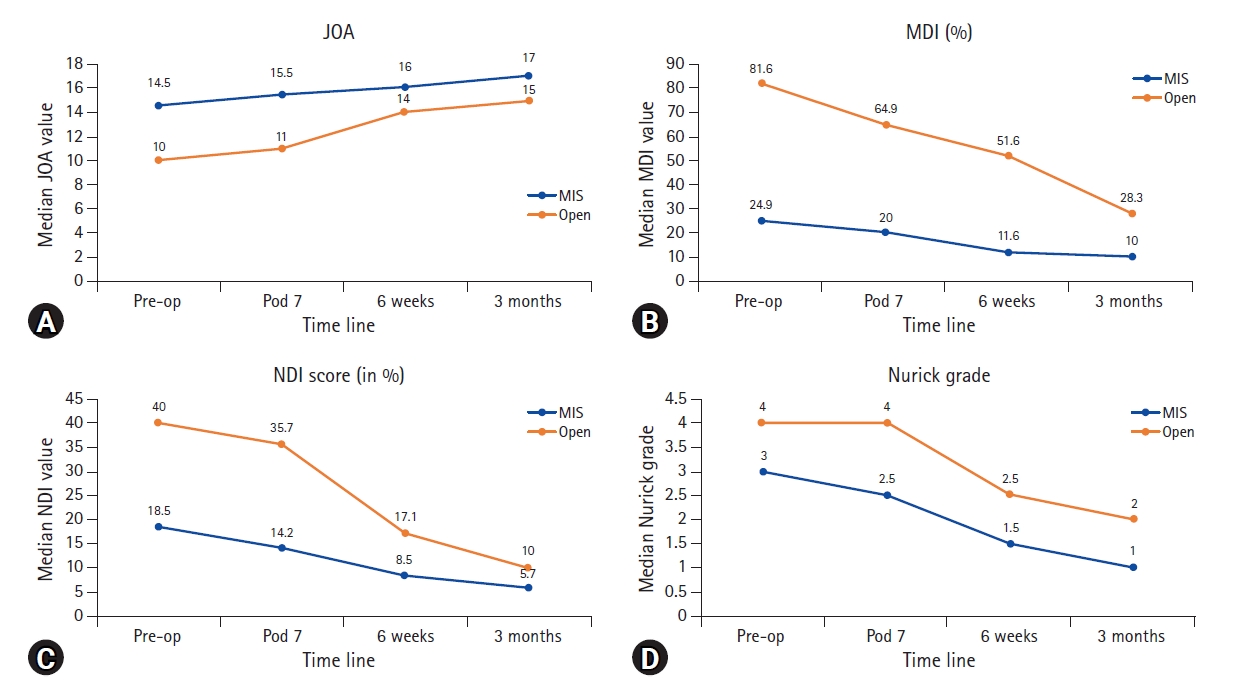
Figure 9.Line charts illustrating the comparison of pre-operative and post-operative values between Open and MIS groups with respect to CSA of dural sac (A) and spinal cord (B). 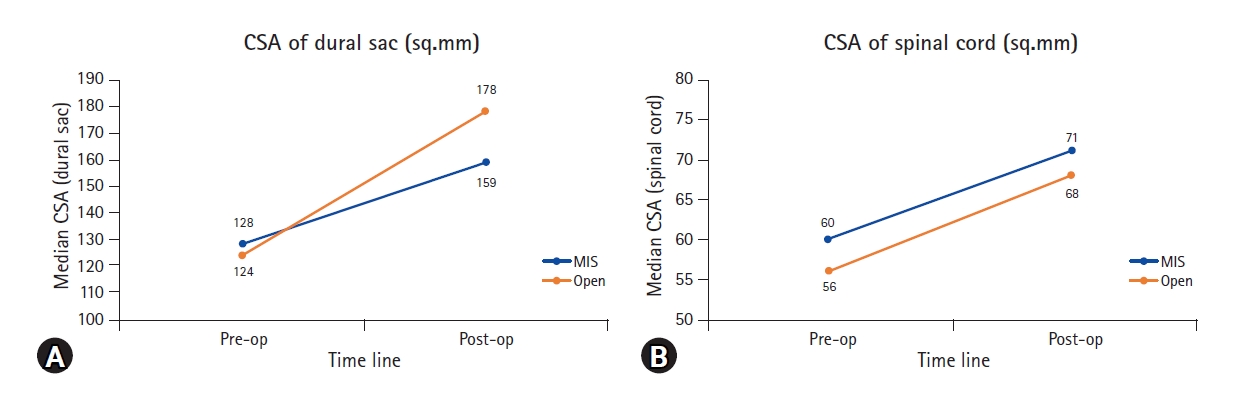
Figure 10.Line charts illustrating the comparison of pre-operative values and post-operative trends at various time points between Open and MIS groups with respect to laboratory parameters such as TLC (A), CRP (B), ESR (C), and CPK (D). 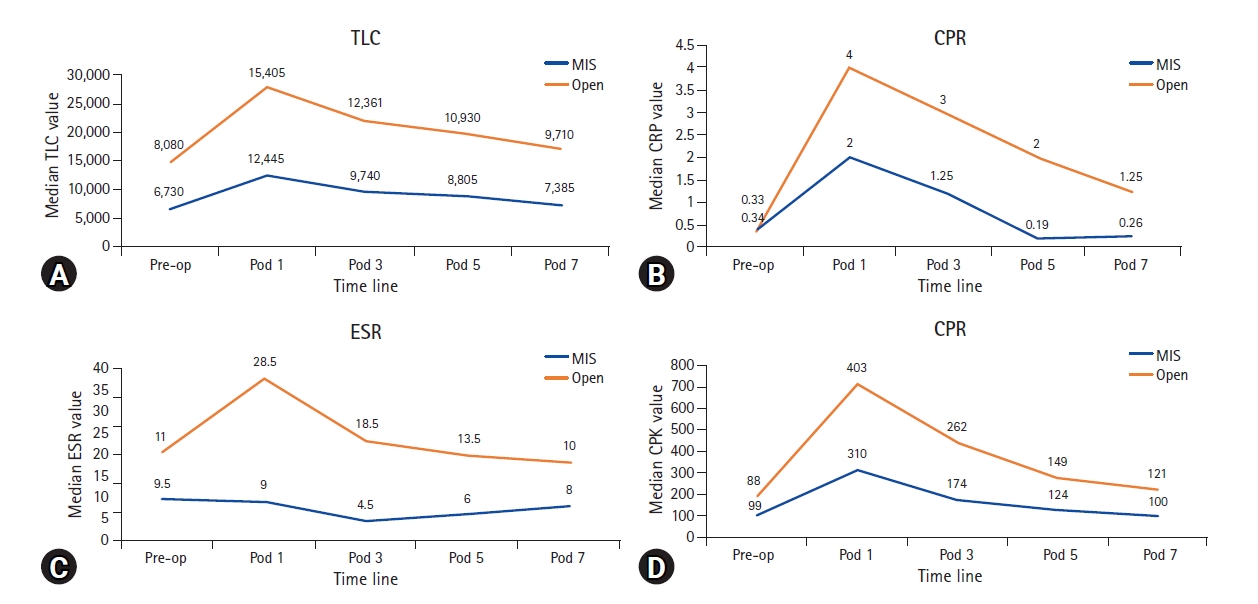
Figure 11.Line chart illustrating the comparison of pre-operative and post-operative values between Open and MIS groups with respect to Isometric neck extensor muscle strength. 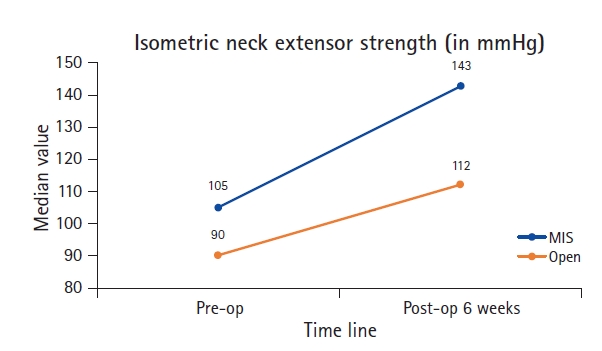
Table 1.Results of clinical and demographic data and comparison between open and MIS groups Table 2.Results of radiological parameters considered for analysis and comparison between open and MIS groups Table 3.Results of post-operative signal changes on T2W axial MRI sequence and comparison between open and MIS groups Data figures represent median values with inter-quartile variations (in parenthesis) of the degree/extent of signal changes graded as mentioned in methods section. The degree of signal changes has been measured at each level. For eg 2-3 is at C2-3 level. PSM: Paraspinal muscle superficial group, SSC: Semispinalis cervicis (deep group). Table 4.Results of laboratory values and comparison between Open and MIS groups Table 5.Results of isometric extensor neck muscle strength (measured with a biofeedback device) and comparison between Open and MIS groups REFERENCES1. Wang SJ, Jiang SD, Jiang LS, Dai LY. Axial pain after posterior cervical spine surgery. a systematic review. Eur Spine J 2011;20:185–194.
2. Ono A, Tonosaki Y, Numasawa T, Wada K, Yamasaki Y, Tanaka T, et al. The relationship between the anatomy of the nuchal ligament, postoperative axial pain after cervical laminoplasty. cadaver and clinical study. Spine (Phila Pa 1976) 2012;37:E1607–E1613.
3. Cho CB, Chough CK, Oh JY, Park HK, Lee KJ, Rha HK. Axial neck pain after cervical laminoplasty. J Korean Neurosurg Soc 2010;47:107–111.
4. Kotani Y, Abumi K, Ito M, Sudo H, Takahata M, Ohshima S, et al. Minimum 2-year outcome of cervical laminoplasty with deep extensor muscle-preserving approach. impact on cervical spine function and quality of life. Eur Spine J 2009;18:663–671.
5. Lee YS, Lee S, Ko MJ, Cho DC, Kim KT. Preservation of deep cervical extensor muscle volume. comparison between conventional open-door and muscle preserving laminoplasty approaches in the same patients. World Neurosurg 2020;141:e514–e523.
6. Shiraishi T, Kato M, Yato Y, Ueda S, Aoyama R, Yamane J, et al. New techniques for exposure of posterior cervical spine through intermuscular planes and their surgical application. Spine (Phila Pa 1976) 2012;37:E286–E296.
7. Kato M, Nakamura H, Konishi S, Dohzono S, Toyoda H, Fukushima W, et al. Effect of preserving paraspinal muscles on postoperative axial pain in the selective cervical laminoplasty. Spine (Phila Pa 1976) 2008;33:E455–E459.
8. Takeuchi K, Yokoyama T, Aburakawa S, Saito A, Numasawa T, Iwasaki T, et al. Axial symptoms after cervical laminoplasty with C3 laminectomy compared with conventional C3-C7 laminoplasty. a modified laminoplasty preserving the semispinalis cervicis inserted into axis. Spine (Phila Pa 1976) 2005;30:2544–2549.
9. Takeuchi T, Shono Y. Importance of preserving the C7 spinous process and attached nuchal ligament in French-door laminoplasty to reduce postoperative axial symptoms. Eur Spine J 2007;16:1417–1422.
10. Rahman M, Summers LE, Richter B, Mimran RI, Jacob RP. Comparison of techniques for decompressive lumbar laminectomy. the minimally invasive versus the “classic” open approach. Minim Invasive Neurosurg 2008;51:100–105.
11. Fan S, Hu Z, Zhao F, Zhao X, Huang Y, Fang X. Multifidus muscle changes, clinical effects of one-level posterior lumbar interbody fusion. minimally invasive procedure versus conventional open approach. Eur Spine J 2010;19:316–324.
12. Tian NF, Wu YS, Zhang XL, Xu HZ, Chi YL, Mao FM. Minimally invasive versus open transforaminal lumbar interbody fusion. a meta-analysis based on the current evidence. Eur Spine J 2013;22:1741–1749.
13. Santiago P, Fessler RG. Minimally invasive surgery for the management of cervical spondylosis. Neurosurgery 2007;60:S160–S165.
14. Celestre PC, Pazmiño PR, Mikhael MM, Wolf CF, Feldman LA, Lauryssen C, et al. Minimally invasive approaches to the cervical spine. Orthop Clin North Am 2012;43:137–147, x.
15. Song JK, Christie SD. Minimally invasive cervical stenosis decompression. Neurosurg Clin N Am 2006;17:423–428.
16. Boehm H, Greiner-Perth R, El-Saghir H, Allam Y. A new minimally invasive posterior approach for the treatment of cervical radiculopathy. surgical technique and preliminary results. Eur Spine J 2003;12:268–273.
17. Hur JW, Kim JS, Shin MH, Ryu KS. Minimally invasive posterior cervical decompression using tubular retractor. the technical note and early clinical outcome. Surg Neurol Int 2014;5:34.
18. Hernandez RN, Wipplinger C, Navarro-Ramirez R, Soriano-Solis S, Kirnaz S, Hussain I, et al; Ten-step minimally invasive cervical decompression via unilateral tubular laminotomy. technical note and early clinical experience. Oper Neurosurg (Hagerstown) 2020;18:284–294.
19. Abbas SF, Spurgas MP, Szewczyk BS, Yim B, Ata A, German JW. A comparison of minimally invasive posterior cervical decompression and open anterior cervical decompression and instrumented fusion in the surgical management of degenerative cervical myelopathy. Neurosurg Focus 2016;40:E7.
|
|
||||||||||||||||||||||||||||||||||||||||